- Joined
- Dec 22, 2007
- Messages
- 526
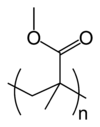
I've never read anything that specifically talks about copper and acrylic tanks. But just looking a the chemical structure of the acrylic, Poly(methyl methacrylate) or PMMA, there is an oxygen double bonded to carbon, a carbonyl group, on the side chain of the monomer. The oxygen of a carbonyl group is slightly negative because it pulls the bonded electrons closer to itself and electrons have a negative charge. A Cu2+, copper ion, would be attracted to this and could move around in the material by popping from one oxygen to another. I may have to ask my o-chem professor what he thinks about it.
interresting thing in the wiki entry on PMMA:
"PMMA has poor resistance to solvents, as it swells and dissolves easily. It also has poor resistance to many other chemicals on account of its easily hydrolyzed ester groups."
interresting thing in the wiki entry on PMMA:
"PMMA has poor resistance to solvents, as it swells and dissolves easily. It also has poor resistance to many other chemicals on account of its easily hydrolyzed ester groups."


 lol. I guess I will wait for a more lamens term for that one..
lol. I guess I will wait for a more lamens term for that one.. 