(continued)
OCTOPOD SUCKER MORPHOLOGY
Functional Morphology:
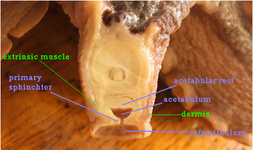
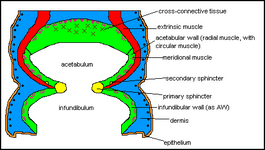
Octopod suckers are very much identical save for a few special cases. They are also functionally and physically different from decapod suckers. The most obvious difference is the lack of a hard structure in the octopus' sucker. Since octopuses use their suckers for a wider range of activities than decapods, such as locomotion, adhering to substratum and even picking up, manipulation and collecting attractive objects, they are more tactile. The basic design revolves around two chambers, as described by Kier and Smith (1990 and 2002), the acetabulum and the infundibulum as shown in fig.7 and fig.8.

fig. 7: the cross section of a sucker from the Eledone cirrhosa specimen. The structure is typical of octopuses, and the only distinguishing characteristic is that this species only has one row of suckers as opposed to 2.

fig. 8: diagram of an octopus sucker distinguishing between the tissue types.
The principles of octopod suckers are similar to that of decapods: in order for the structure to adhere to an object, the aim is to generate a reduced pressure. While squids and cuttlefish employ the use of a muscular piston, octopus suckers use two cavities. A seal is formed between the object and the sucker, by the fleshy rim. The radial muscles in the acetabular wall and roof contract so that the acetabulum increases in size. This is because the radial wall and roof become thinner and longer making the acetabulum wider and taller. If this were to happen in the water column then water would just rush in to fill the larger area. However, the sucker is pressed against an object, prey item or piece of substratum, so no excess water can enter. The water inside the sucker resists expansion and so there is a decrease in pressure. This water essentially begins to act like a solid whilst under pressure, allowing adherence to the surface. In the acetabular wall, the circular muscle bundles are the antagonists of the radial muscle, and reduce the circumference and height of the acetabulum upon contraction, which in turn releases the sucker.
Structural Morphology:
As I have already described, octopus suckers do not show much variation, save in a few very specialised cases. It seems to be that generalisation limits the variation in structure, whereas specialisation encourages unique features at the expense of how many functions it can perform. Although the basic octopus sucker doesn't vary much across species, except for maybe slight superficial changes, such as size and shape, it can perform many extraordinary feats such as being able to literally fold in half in order to hold onto an object as thin as a sheet of paper (Kier and Smith 2002)! Octopus suckers are also able to adhere to irregular surfaces, presumably due to them not having hard parts anywhere. They can also be used as chemotactile organs (Kier & Smith, and also Chase & Wells, 1985). Octopuses do not just forage using eyesight alone, although they are clearly visually-oriented animals, and frequently search for food using their arms to probe where they can't see. Perhaps a cross between touch and taste (or smell!) would be the best description of the term "chemotactile". Octopuses are also attracted to items and objects in a similar way to magpies. Frequently it will pick up, for example, a piece of glass that is glinting in the sunlight and carry it back to its den where it keeps a collection or "horde" of shells and debris. As well as adhering to the smooth edge, it would probably grasp the thin edge by bending a sucker in close proximity to it. An analogy could be a party game where the participant has to pick up an object using only a folded paper plate.
The arrangement of the suckers is also interesting. In general, the suckers begin very close to the mouth, and increase in diameter while continuing out along the arms in either one (usually associated with the Eledone genus) or two (the more common number) rows before decreasing in diameter as the arm thin out to a point. The difference in the basal diameters is clear in fig.9. The mouth can also be seen in fig 9, which is the opening in the centre of the suckers in the distance.

fig. 9: oral view of E. cirrhosa. Note the distinct increase of sucker diameter. The diameters were relatively constant until quite far past the median in this specimen, only decreasing once they started reaching the tips. Also notice how flexible the suckers look. It is also possible to see the acetabulum through the holes of a few of the suckers.
The suckers can also rotate, and move quite freely (as demonstrated by the example of it folding), which is how the octopus is able to manipulate prey and move it towards the mouth.
Most octopuses have a hectocotylus. This takes the form of a specialised arm for use in reproduction and includes a sperm channel. The hectocotalised arm in incirrate octopods is one of the third arms (Arm 3), and is inserted into the female's mantle cavity to allow transfer of the males' sperm packets.
The lining of the infundibulum, a thin pellicle, is frequently cast off. During this activity the octopus is commonly said to be "shedding its suckers" (Nesis). Presumably this is to ensure that the condition of the infundibulum is kept optimal, and free of dead skin or other debris that could impair its adhesive abilities. Since octopuses spend a great deal of time attached to surfaces that are potentially colonised by algae and other small colonial or film- producing organisms, a good way to keep the sucker free of unwanted hitchers and to ensure reliable function, would be to shed the layer that's in contact with these conditions the most!
Another reason could be that the octopus would want to keeps its suckers as flexible and supple as possible, and if the lining inside gets too rigid or firm, then the sucker will become limited in its movements and its usefulness impaired. Essentially, frequent shedding will keep the pellicle fresh.
PREHISTORIC COLEOIDS: BELEMNOIDEA
Belemnoids are prehistoric coleoids that were most abundant during the Mesozoic era, and became extinct at the end of the Cretaceous. They came from the same ancestral stock as the neocoleoids, and lived alongside them from the early Carboniferous right up until their demise at the KT boundary.
Belemnoids typically display the ancestral arm pattern (fig. 1a), comprising ten subequal arms, with no distinct specialisation. This is very much speculative, since we only know of belemnoids from fossil evidence. There have been some well preserved individuals, however (Fig. 10). Of course they do have hard parts, but even the soft parts, such as the arms, and mantle, have been preserved in the rock remarkably well on rare occasions, possibly implying a very muscular, and not gelatinous, animal. On the other hand it could have just been the composition of the sediment in which the animal came to rest.
A remarkable feature of the belemnoids, and one that fossilises very well, is the presence of distinct, large hooks running in two series along the oral surface of the animal (Engeser & Clarke, 1988). Figs.10 and 11 show photographs of fossilised belemnoid remains.
The basic belemnoid hook probably comprised the same components as that of extant coleoids; namely chitin. It is possible that the hooks contained more components, such as harder, possibly mineral, materials, but then the only evidence for this really is that the hooks are so well preserved, compared to fossil neocoleoid suckers rings. The hook was positioned curving inwards, and in two series running along each arm, in pairs of between 20 and 50.

fig. 10: Acanthoteuthis from Solenhofen, Germany, currently residing in the collection of U. S. National Museum of Natural History. Of particular recognition is the clear detail of the animal's body, including the hooks clearly radiating off the arms!

fig. 11: belemnoid hooks (possibly Belemnoteuthis) taken from Lyme Regis in Dorset, England, also in the collection of U. S. National Museum of Natural History. Both photographs used with kind permission by Richard E. Young, University of Hawaii.
The hook itself is split up into several sections, each with its own distinct nomenclature as shown in fig.13. The presence of this small hairline groove known as the orbicular scar indicates that the hook was probably not exposed, and was housed within a sheath of soft tissue. Because the tissue attachment point is quite far up the shaft, only the uncinus would be visible, or at least naked. Engeser and Clarke proposed that the hook was stalked, and that it was attached by muscles that could have allowed lateral movement. Since the belemnoid did not have an extensile pair of limbs, like the tentacles of squids and cuttlefish, or the flexible, muscular sucker discs like octopuses, it would have had to manipulate the prey for eating by using its whole arm. If the hooks were sessile, bearing in mind that it may have had up to fifty, then the belemnoid would be severely limited in its arms' movement. Having hooks that could be moved, even slightly, would allow the animal more movement. The manner in which the hooks were positioned, recurved, would have also helped, in that the trapped prey, which was obviously caught by being impaled, could only move in one direction in order to free itself from the hook: towards the mouth. According to Engeser and Clarke, there are three groups of belemnoids that display arm hooks, and one that does not. The Phragmoteuthids are reported as far back as the late Permian and are described as having more slender, slighter hooks than what is described as the "normal" or archetypal hook (fig.13) although variations including enlarged bases and more curved uncini are reported in species. The Belemnoteuthids have more typical hooks, although Chondroteuthis wunnenbergi Bode 1933, exhibited prominent spurs on the proximal lower third. The Belemnitids, most well known as the belemnites, also display a deal of variation. Acrocoelites (Toaricibelus) raui Werner 1912 appears to have two different types of hook, differentiating from the typical hook to those with an internal spur towards the distal end of the arm. Two fossil specimens of Passaloteuthis paxillosa Schlotheim 1820 showed the same structural oddities of having no hooks whatsoever towards the distal parts of the arms. This suggests that this species may well have lacked hooks at the ends of their arms. Unfortunately no other specimens were described or even mentioned, so a conclusion can only be drawn from these two fossils alone.

fig. 13: schematic of a typical belemnoid hook, adapted from Engeser & Clarke
Ideally a more thorough investigation into prehistoric cephalopod arm structure would have been desirable, but there is so little fossil evidence of cephalopods, being a soft-bodied animal naturally, that such a task is nigh on impossible at present. Most of the neocoleoid fossils are little more than faint outlines with maybe the presence of one or two organs, or surfaces, but this can be sketchy at best, since the fossils are interpreted differently. For this reason, only belemnoids have been included as a representative of prehistoric cephalopods since the available information on fossil cephalopods with regard to armature is pretty much restricted to the belemnoids. It is curious just how well these animals have preserved compared to the other prehistoric cephalopods, given that this class has been in the world's oceans since the Late Cambrian.
SPECIAL CASES
The case of Stauroteuthis syrtensis:
Stauroteuthis syrtensis Verrill 1879 (Johnsen et al., 1999) is a deep sea cirrate octopod that sports very unique armature. It is similar morphologically to other cirrate octopods in that it possesses two rows of cirri along the oral surface (the underside) of each arm, and also has a long web that connects each arm with a single series of structures analogous to the suckers of a typical octopus such as E. cirrhosa or Octopus vulgaris. However, these structures only vaguely resemble their adhesive counterparts, and can even be described as vestigial. Indeed these suckers no longer perform any adhesive role whatsoever, instead they exhibit Bioluminescence! Although bioluminescence is common among the decapods (Johnsen et al.) it is incredibly rare in octopods, being described in that manuscript as only being present in "2 of the 43 genera", and even then only found in breeding females. In this species, however, it appears that both females and males possess the light organs (photophores). Robison and Young (1981) observed that the light emitted from these modified suckers were of a definite green hue, compared to the blue-green light more commonly found in bioluminescent animals. This does set it apart from other inhabitants of the deep with such similar abilities!
The problem with the information regarding this species, as pointed out by the team that produced said document, is that it lives at great depths. Indeed the three specimens were caught in a range of 750- 920m using deep trawling methods. They are understandably difficult to study in their natural environment not only due to the depth, or the mind-blowing vastness of its habitat, but also because the octopod can probably detect the submersible light long before contact, meaning that it is unclear whether the behaviour observed is natural or induced by the presence of the submersible. It is described as hanging in the water column with its arms and web outstretched to create a sort of bell-shaped posture. It is possible that this is a threat response, but it appears to be a natural position rather than that of defence. There are a few theories as to why such an animal would develop bioluminescence, which can be both an advantage and a disadvantage, as will be discussed.
S. syrtensis exhibits sexual dimorphism in that mature males have grossly enlarged suckers (or photophores) compared to those of a female of similar development. Since both have the bioluminescent ability, it was proposed that it was used as sexual displays. Sexual signalling is understandably common in a phylum that appears to rely so much on vision, and so this theory would appear to make sense. The evidence of a distinct light colour also enables what Johnsen, Balser, Fisher and Widder described as a "private line of communication" between members of the same species. Indeed, any clear visual cue that distinguishes you from another species, especially in the dark, is bound to be favourable! However, there are a few factors that may serve to contradict this theory. The first is that in this instance, all three specimens examined had these photophores, and yet they all appeared to be immature. Surely only mature individuals, with the ability to reproduce, would only need these? But then, we only know a small amount about this species, so anything is possible. Another problem is that the octoradial pattern (imagine eight twinkling spokes) would be distorted after a certain distance due to light dispersal. However, it is questionable that this would prove too much a problem since the photophores emit a fairly distinct green colour! Finally this colour would be clearly visible to potential predators. That said, although in a slightly different context, many male birds compromise cryptic camouflage in favour of conspicuous coloration and displays that give its presence away, in order to attract mates. Although sexual signalling probably isn't the primary reason for photophores, I am not, at such an early stage, wholly willing to dispel the possibility.
This being the case, a slightly more plausible hypothesis describes the light's use in prey capture. S. syrtensis feeds on small, planktonic crustaceans. It has been proposed that cirrate octopods secrete lines of mucous web which are handled by the cirri to trap the animal's prey. This kind of method has been suggested for three of the cirrate genera (all of which possess "non-functional" suckers), Stauroteuthis being one of them. In this case, the bell posture could be easily explained. It is possible, therefore, that the photophores are used as a lure, similar to the angler fish. These crustaceans have well developed eyes, and seem to be attracted to light sources, which would serve the S. syrtensis very well. The fact that the animal is usually observed in the bell or umbrella posture either oriented with its mouth facing upwards, or facing downwards could coincide with nutrient flows in the water column, since many planktonic and nektonic animals make vertical migrations every night in order to feed on phytoplankton. On the other hand it could just be, as Denton (1990) stated, because upwelling light intensity is a small percentage of downwelling light intensity, which Johnsen et al. proclaimed would make the octopod highly visible to potential prey higher up in the water column, although this statement I have difficulty in imagining. Surely if the light intensity of an octopod facing upwards was less it would be less visible to prey above it?
The case of the Vampyromoprh:
The Vampire squid, Vampyroteuthis infernalis Chun 1903 is the only known remaining member of the order Vampyromorpha (sometimes Vampyromorphida). There are fossils of these animals ranging back to the Mesozoic, which is rather remarkable given their gelatinous tissue composition! A resident of the deep oceans, this is yet another relatively unknown animal.
The vampire squid, although commonly referred to as a "squid", is a member of the Octopodiforme superorder. Its second arms (Arms 2; the ones octopods have lost) have been modified to retractile sensory filaments that actually look like they are not part of the limb structure since they are not connected by the web. Like the cirrates they only possess one series of suckers, which are "banked" by a row of cirri on either side, and in V. infernalis are only present of the distal half. The web is attached to the arms to about two thirds of the way.
Young (1967) showed that the filaments are innervated differently to the other eight arms, which suggests that they are derived from a source different to the "decapod ancestor". These filaments are used in feeding and are probably used like a fishing line. A prey item bumps into the filament in the dark waters. Upon sensing this stimulus, the vampire squid lunges toward the source in the hope of enveloping it with the arms and web. It is possible that the cirri play a part in this too. The vampire squid also displays unique behaviours, especially in response to a potential threat. One threat response is to invert its arms and web round its head, as if it's turning itself "inside out", and extending its cirri outwards so that they resemble spikes or hooks (fig.14) in what is known as a "Pineapple posture".

fig. 14: photograph of the vampire squid in its "pineapple" posture with cirri held rigid.
Another predator-avoidance strategy is the use of photophores in the skin, combined with ejection of mucous containing bubbles of bioluminescent light. Once a string has been completed the squid halts operation of its photophores and slips of, leaving the bioluminescent mucous thread decoy with the predator.
The suckers of V. infernalis lack a cuticular lining, which may suggest that they do not have the ability to seal the sucker to an object, meaning the sucker would be useless, if not extremely weak, as an adhesive structure.
The case of the Nautiluses:
The nautiloids are the oldest cephalopods on the planet. From the Cambrian to the Early Devonian they were the only cephalopods found in the oceans, and even continued to dominate right through to the Jurassic period, where they experienced a sharp decline, which saw the extinction of the orthoconic nautiloids. It is the early nautilda that gave rise to our modern nautiluses, of which there are only around six known species. The early nautilda showed many of the characteristics familiar with these modern species. The nautilus really is a relic of an ancient time!
What sets the nautiluses apart from the coleoid cephalopods is the absence of suckers. By definition, nautiluses do not have arms, and instead can have anywhere up to ninety tentacles which are kept in sheaths when not in use. The absence of suckers does not dictate a lack of adhesive ability however, and the nautilus still employs special structures to allow it to grip objects, albeit using a completely different strategy.
Nautiluses have, lining their tentacles, a series of alternating ridges, or cirri, and grooves. These allow nautiluses to grip substrata or prey with an effect similar to Velcro. In the emperor nautilus, Nautilus pompilius Linnaeus 1758, and most likely in the other five species (and both N. pompilius subspecies), the cirri are in turn lined with annular ridges that run round the circumference of each cirrus, and are more pronounced on the oral side (Muntz & Wentworth, 1995). The epithelium exhibits thickening on the proximal surface of these ridges, and houses electron-dense granules which are only found in this location on the cirrus. The granules contain a mucopolysaccharide which could be what facilitates adhesion.
Nautiluses have incredibly poor vision; they're eyes being little more than pinhole eyes (Basil et al., 2000), meaning that they can probably only distinguish light and dark, and are most likely unable to clearly define objects. Clearly nautiluses do not rely on vision to hunt or scavenge; they tend to feed on dead matter but they are still active predators so they need to forage, and locate prey.
Their tentacles also house cells that resemble taste buds (Basil et al.), but were found not to allow the animal to track the odour, or chemical signal. For this the animal requires two specialised organs located below the eye called Rhinophores. While the tentacles were found to pick up chemicals in the water, it was the rhinophores that allowed them to locate the source. Test animals with their rhinophores temporarily plugged did show a distinct behavioural change upon the presence of a chemical in the water, but they could not effectively locate the source. This suggests that the tentacles could pick up preliminary stimuli which tell the animal that a food source is there. The emperor nautilus inhabits fairly deep regions (300m approx.) and migrates vertically at night to feed. Essentially it lives in a dimly-lit, or even completely dark, world. If the animal was using its rhinophores all the time, such a behaviour would surely be wasteful, and so a preliminary warning system that tells the animal when it can use its rhinophores to locate prey would be a huge benefit.
Aside from foraging, the nautilus also uses its tentacles to adhere to surfaces during stages of inactivity. According to O'Dor, 2002, nautiluses engage in a state of torpor akin to sleep. He also stated that an animal in this state, if not anchored to any substratum, would drift around at a velocity fairly close to its optimum speed, in a random fashion. This movement is induced by breathing alone. Since nautiluses achieve locomotion by "jetting". Since they move by expelling water from their mantle cavity, the same site where the gills are situated, random movement would be understandable during unconsciousness. This of course poses many hazardous implications for the nautilus, and the animal would quite rightly want to stay put!
CONCLUSION
Similarities and Differences:
The sucker of the decapods and octopods are analogous. They both perform exactly the same function; adherence to a surface, but the morphology is completely different. The basic uses are also the same, although the octopods tend to have more uses for their sucker pads.
It would appear that decapod suckers are very limited in their uses, being mostly for prey capture: to aid in the adherence of the sucker to the body of the prey, and in some cases, probably by impalement! Some hook structures, such as those found in sexually dimorphic species, especially in those in which the dimorphism is dictated by the hooks, there is the possibility of a use in reproduction, most likely in species that reproduce through direct implantation by the male of sperm into the female's mantle cavity. Aside from this, decapod suckers do not appear to have any other role. Octopuses use their suckers for more than just prey capture and manipulation, and the suckers are more flexible and muscular. The sucker structure itself is relatively sessile, but the rim of the infundibulum has a remarkable array of movement. Octopuses are regarded as highly intelligent animals, which could be a result of their multipurpose suckers, or even vice versa. The intelligence of decapods was not researched for this paper, but it is possible that squids at least cannot achieve the intellectual level of an octopus, on the evidence of their highly specific armature. This statement is completely subjective, however, but it is one conclusion that can be drawn from the observation of the different structures.
The decapods do show a lot more variety in terms of structure, such as the shape and size of the ring in relation to the sucker. This may be a reflection on the animal's prey. Since nearly every family, at least, have architectural differences in the ring the different shapes could prove optimal formations for catching particular prey species, or perform better at different velocities or depths. In a three-dimensional world, it would also be wise to include the decapod's angle in relation it its prey, which might also affect the success of the tentacle strike. Decapods typically have a very fast tentacle strike; the tentacle extending and contacting a prey item in about 15-35ms (Smith, referring to Kier, 1982), with an incredibly high success rate: normally 80-90%. The chitinous rings could play a large part in this success, allowing an initial grip which facilitates formation of a rim, in turn allowing adhesion by pressure differential. Owing to the highly specialised use of tentacles, it is possible that decapods do not have as much motor control over their tentacles as they do their arms.
Octopus capture methods may differ somewhat as the octopus is using one or more of its arms to grab a prey item, which it probably has more control over, compared to a pair of tentacles. As such it can probably allow for a much surer strike. Octopus arms also frequently coil up when they grab an object, which no doubt increases its hold over the item, which means that they may not require hard, claw-like formations. Octopuses also lunge at their prey with all their arms, usually from above, in the water column. This behaviour is also demonstrated by cuttlefish. Another reason why octopuses do not require hooks or rings is that they are ambush predators and are masters of camouflage, able to change their appearance and colour at will. This means that they can close in on a prey animal, and will only strike when they are sure of success. In a three-dimentional environment, such as those of pelagic squids, there is no such luxury of a complete surprise attack, and so the squid would have to rely more on a high-velocity mode of capture.
Indeed, hard parts may even prove detrimental to an octopus, given the majority of octopuses' habitats; the sea floor. Octopuses live in and around rocks, and frequently reside in dens. It is common knowledge of the octopus' ability to squeeze into the narrowest gaps, and its inquisitiveness and tendency to explore and probe. An octopus can go anywhere so long as its beak can get through. If an octopus was carrying around so many hard structures in its arms, then it would be severely limited in where it would go.
It seems to be the case generalisation demands a simpler anatomical structure (in this case the octopod's suckers) whereas specialisation begins to limit the range of uses, in order for that structure to be optimal in that one special activity. This appears to also be the case in cephalopods with regards to their structures.
Cephalopods are very visually oriented animals, and employ the use of complex signals. This does include very complex, and impressive colour displays using chromatophores to switch to a variety of colours to beautiful effect, but the use of arm signals is also important. Although this is more holistic, and may not even include the suckers in some cases, an interesting implication does arise when considering sexual dimorphism. As in the case of L. grimaldii it was presumed that the hooks were used physically in mating, but just as probable is role in display. Just as stags sport their impressive antlers during the rut, that are usually only employed as a grappling weapon if two competing males refuse to back down, so the male scaled squid may perform similar displays to impress a female. Until two scaled squid are found in the act of mating, we can only speculate.
A conclusion can also be drawn from the terminology derived from the observation of cephalopod limbs, namely the tentacles. It is found that in both the coleoid decapods and the nautilus both have limbs called tentacles. Which it was observed that tentacles were specialised limbs, what is consistent in both subclasses is that these limbs are fully retractable and extendible. Indeed nautiluses only sport this kind of limb, and do not have any unspecialised arm. Whether it was by coincidence, it can nevertheless possible that the term tentacle can refer to a limb which is extendable.
A possible Evolutionary Theory:
The existing Nautiloidea, of the genus Nautilus, exhibit what could be classed as a rather primitive array of armature. Providing the Nautiloidea did possess these adhesive ridges to begin with, and it wasn't just convergent evolution displayed by the nautiluses, this could very well explain the beginnings of the cephalopod sucker structure. It's doubtful that nautiluses alone would just happen to develop an adhesive system on their limbs in recent times without good reason, and so it would be expected that the ridges are the very ancestral progenitor of the sucker pad. This theory would be ideally backed up by the discovery of soft-bodied bactritid ammonoid fossils, these being the intermediary stage between nautiloidea and coleoidea, and ancestral to the later ammonoids with perfectly preserved arms that could show whether any structures were present. Since in ammonoids the only fossils reported have thus far only been the shell, conclusions have to be drawn.
Assuming that the nautiloids did have adhesive ridges, it can be suggested that these were carried and further developed by the ammonoidea: more specifically the aforementioned bactritids, which gave rise to the coleoids.
Assuming also that the bactritids (at least) had some sort of ancestral structure, such as a hook, makes the presence of the belemnoid hook more plausible as well as the neocoleoid hook. These structures could not have just sprung up out of nowhere.
It is possible, therefore, that the hook structure was possessed by a common ancestor to belemnoids and neocoleoids. This common ancestor could have had an ancestral hook. When the belemnoids and neocoleoids diverged, the belemnoids may have kept the ancestral hook (or indeed developed it in a completely different way!), while the neocoleoids developed the hook into a sucker pad. A short diagram showing the development from hook to sucker is shown in fig. 15.

fig. 15: a depicts development from an ancestral hook similar to that of a belemnoid's to that of a modern decapod, including the formation of the cavity (acetabulum) and a stalk, and the transition from a single hook to a ring of denticles. Included is b which depicts what could happen, carrying on from a in the same direction.
For sake of completeness, further development into the octopods sucker, lacking a ring, is included. If this were to happen, then because octopods did not branch off of decapods, and both more or less diverged in the same manner as with the belemnoids, theoretically the first octopods would have had some hard structures until these vanished. Of course the internal morphology would have to change again, but slight modification of the infundibulum and acetabulum, and the development of the piston into the acetabular roof could facilitate this. This is highly speculative as there is not a lot of evidence of fossil neocoleoids, but I'd like to hope that it fits together as a sound theory. One interesting fact is that the "paper nautilus" Argonaut argo, an incirrate octopus that secretes a very thin shell, appears to have hooks between the rim of the sicker and infundibulum (Nixon & Dilly); could this be a throwback to a day when all coleoid cephalopods had hard armature?
Thanks:
I would like to thank the following people for their advice and help throughout the duration of my study, in no particular order:
I thank the display manager at St. Andrews Aquarium for the specimen of Eledone cirrhosa as well as giving advice and showing an interest in the study. I also thank William Kier for providing me with his two papers on octopus suckers and having an interest from the offset.
Special thanks goes to the staff and forum members of TONMO, for the endless help, ideas, advice and critical views, including Phil Eyden for proof reading this paper and providing extensive information on fossil cephalopods, Dr Steve O'Shea of Auckland University of Technology in New Zealand, and especially Kat Bolstad, also of Auckland University, who provided endless help and sources. I would have struggled to obtain some sources otherwise.
Finally, within the department, I would also like to thank several people. Obviously I would like to thank my supervisor, Dr Allan Jones, for suggesting the topic that has dominated my life for the last half year, as well as providing valuable advice and moral support. Also of note is the university library, who managed to negotiate an extended inter-library loan of "Cephalopods of the World" for me. I would also like to thank a fellow student, Peter Rendle, for posing very interesting questions, as well as good, in-depth discussion regarding the subject.
REFERENCES
Anderson F. E. 2000, Phylogenetic relationships among loliginid squids (Cephalopoda: Myopsidia) based on analyses of multiple data sets. Zoological Journal of the Linnean Society 130: 603-633
Bolstad K. S., O'Shea S. 2004, Gut contents of a giant squid Architeuthis dux (cephalopoda: Oegopsida) from New Zealand waters. New Zealand Journal of Zoology 31: 15-21
Chase r., Wells M. J. 1986, Chemotactic behaviour in Octopus. Journal of Comparative Physiology 158/3: 375-381
Christensen W. K. 2002, Fusiteuthis polonica, a rare and unusual belemnite from the Maastrichtian. Acta Palaeontologica Polonica 47: 679-283
Dzik J. 1980, Origin of the cephalopoda. Acta Palaeontologica Polonica 26: 161-191
Engeser T. S., Clarke M. R. 1988, Cephalopod hook, both recent and fossil. The Mollusca 12: 133-151
Fiorito G., Gherardi F. 1999, Prey-handling of Octopus vulgaris (Mollusca, Cephalopoda) on Bivalve preys. Bejhavioural Processes 46: 75-88
Förch E. C., Uozumi Y. 1990, Discovery of a specimen of Lycoteithis lorigera (Steenstrup, 1875) (Cephalopoda: Teuthiodea) from New Zealand and Additional notes on its morphology. New Zealand Journal of Marine and Freshwater Research 24: 251-258
Jackson G. D., O'Shea S. 2003, Unique hooks in the male scaled squid Lepidoteuthis grimaldii Joubin 1895. Unpublished but available through the TONMO website
Johnsen S., Balser E. J., Fisher E. C., Widder E. A. 1999, Bioluminescence in the deep-sea cirrate octopod Stauroteuthis syrtensis (Mollusca: Cephalopoda). Biological Bulletin 197: 26-39
Keir W. M., Smith A. M. 1990, The morphology and mechanics of octopus suckers. Biological Bulletin 178: 126-136
Keir W. M., Smith A. M. 2002, The structure and adhesive mechanism of octopus suckers. Integrated Comparative Biology 42: 1146-1153
Kier W. M., Van Leeuwen J. L. 1997, A kinematic analysis of tentacle extension in the squid Loligo pealei. The Journal of Experimental Biology 200: 41-53
Lindgren A. R., Giribet G., Nishiguchi M. K. 2004, A combined approach to the phylogeny of Cephalopoda (Mollusca). Cladistics 20: 454-486
Lipinski M. R. 2001, Preliminary description of two new species of cephalopods (Cephalopoda: Brachioteuthidae) from South Atlantic and Antarctic waters. Bulletin of the Sea Fisheries Institute 1 (152) : 3-14
Lukeneder A. 2005, First nearly complete skeleton of the Cretaceous duvaliid belemnite Conobelus. Acta Geologica Polonica 2: 147-162
Muntz W. R. A., Wentworth S. L. 1995, Structure of the adhesive surface of the digital tentacles of Nautilus pompilius. Journal of the Marine Boilogical Association of the United Kindom 75 (3): 747-750
Nixon M., Dilly P. N. 1977, Sucker surfaces and prey capture. Symposium, Zoological Society, London 38: 447-511
Robison B. H., Young R. E. 1981, Bioluminescence in pelagic octopods. Pac. Sci. 35: 39-44
Smith A. M. 1996, Cephalopod sucker design and the physical limits to negative pressure. The Journal of Experimental Biology 199: 949-958
Vecchione M., Galbraith J. 2001, Cephalopod species collected by deepwater exploratory fishing off New England. Fisheries Research 51: 385-391
Nesis K. 1987, Cephalopods of the World. Originally published in Russia, 1982, this translated edition T.F.H Publications ISBN 0-86622-051-8
Tree of Life web project: www.tolweb.org/tree
Cephbase: www.cephbase.utmb.edu
Waikiki Aquarium website: www.waquarium.org
The Cephalopod Page: www.thecephalopodpage.org
Paleos: www.palaeos.com
All sites are operational as of July 2007.
Functional Morphology:
Octopod suckers are very much identical save for a few special cases. They are also functionally and physically different from decapod suckers. The most obvious difference is the lack of a hard structure in the octopus' sucker. Since octopuses use their suckers for a wider range of activities than decapods, such as locomotion, adhering to substratum and even picking up, manipulation and collecting attractive objects, they are more tactile. The basic design revolves around two chambers, as described by Kier and Smith (1990 and 2002), the acetabulum and the infundibulum as shown in fig.7 and fig.8.

fig. 7: the cross section of a sucker from the Eledone cirrhosa specimen. The structure is typical of octopuses, and the only distinguishing characteristic is that this species only has one row of suckers as opposed to 2.

fig. 8: diagram of an octopus sucker distinguishing between the tissue types.
The principles of octopod suckers are similar to that of decapods: in order for the structure to adhere to an object, the aim is to generate a reduced pressure. While squids and cuttlefish employ the use of a muscular piston, octopus suckers use two cavities. A seal is formed between the object and the sucker, by the fleshy rim. The radial muscles in the acetabular wall and roof contract so that the acetabulum increases in size. This is because the radial wall and roof become thinner and longer making the acetabulum wider and taller. If this were to happen in the water column then water would just rush in to fill the larger area. However, the sucker is pressed against an object, prey item or piece of substratum, so no excess water can enter. The water inside the sucker resists expansion and so there is a decrease in pressure. This water essentially begins to act like a solid whilst under pressure, allowing adherence to the surface. In the acetabular wall, the circular muscle bundles are the antagonists of the radial muscle, and reduce the circumference and height of the acetabulum upon contraction, which in turn releases the sucker.
Structural Morphology:
As I have already described, octopus suckers do not show much variation, save in a few very specialised cases. It seems to be that generalisation limits the variation in structure, whereas specialisation encourages unique features at the expense of how many functions it can perform. Although the basic octopus sucker doesn't vary much across species, except for maybe slight superficial changes, such as size and shape, it can perform many extraordinary feats such as being able to literally fold in half in order to hold onto an object as thin as a sheet of paper (Kier and Smith 2002)! Octopus suckers are also able to adhere to irregular surfaces, presumably due to them not having hard parts anywhere. They can also be used as chemotactile organs (Kier & Smith, and also Chase & Wells, 1985). Octopuses do not just forage using eyesight alone, although they are clearly visually-oriented animals, and frequently search for food using their arms to probe where they can't see. Perhaps a cross between touch and taste (or smell!) would be the best description of the term "chemotactile". Octopuses are also attracted to items and objects in a similar way to magpies. Frequently it will pick up, for example, a piece of glass that is glinting in the sunlight and carry it back to its den where it keeps a collection or "horde" of shells and debris. As well as adhering to the smooth edge, it would probably grasp the thin edge by bending a sucker in close proximity to it. An analogy could be a party game where the participant has to pick up an object using only a folded paper plate.
The arrangement of the suckers is also interesting. In general, the suckers begin very close to the mouth, and increase in diameter while continuing out along the arms in either one (usually associated with the Eledone genus) or two (the more common number) rows before decreasing in diameter as the arm thin out to a point. The difference in the basal diameters is clear in fig.9. The mouth can also be seen in fig 9, which is the opening in the centre of the suckers in the distance.

fig. 9: oral view of E. cirrhosa. Note the distinct increase of sucker diameter. The diameters were relatively constant until quite far past the median in this specimen, only decreasing once they started reaching the tips. Also notice how flexible the suckers look. It is also possible to see the acetabulum through the holes of a few of the suckers.
The suckers can also rotate, and move quite freely (as demonstrated by the example of it folding), which is how the octopus is able to manipulate prey and move it towards the mouth.
Most octopuses have a hectocotylus. This takes the form of a specialised arm for use in reproduction and includes a sperm channel. The hectocotalised arm in incirrate octopods is one of the third arms (Arm 3), and is inserted into the female's mantle cavity to allow transfer of the males' sperm packets.
The lining of the infundibulum, a thin pellicle, is frequently cast off. During this activity the octopus is commonly said to be "shedding its suckers" (Nesis). Presumably this is to ensure that the condition of the infundibulum is kept optimal, and free of dead skin or other debris that could impair its adhesive abilities. Since octopuses spend a great deal of time attached to surfaces that are potentially colonised by algae and other small colonial or film- producing organisms, a good way to keep the sucker free of unwanted hitchers and to ensure reliable function, would be to shed the layer that's in contact with these conditions the most!
Another reason could be that the octopus would want to keeps its suckers as flexible and supple as possible, and if the lining inside gets too rigid or firm, then the sucker will become limited in its movements and its usefulness impaired. Essentially, frequent shedding will keep the pellicle fresh.
PREHISTORIC COLEOIDS: BELEMNOIDEA
Belemnoids are prehistoric coleoids that were most abundant during the Mesozoic era, and became extinct at the end of the Cretaceous. They came from the same ancestral stock as the neocoleoids, and lived alongside them from the early Carboniferous right up until their demise at the KT boundary.
Belemnoids typically display the ancestral arm pattern (fig. 1a), comprising ten subequal arms, with no distinct specialisation. This is very much speculative, since we only know of belemnoids from fossil evidence. There have been some well preserved individuals, however (Fig. 10). Of course they do have hard parts, but even the soft parts, such as the arms, and mantle, have been preserved in the rock remarkably well on rare occasions, possibly implying a very muscular, and not gelatinous, animal. On the other hand it could have just been the composition of the sediment in which the animal came to rest.
A remarkable feature of the belemnoids, and one that fossilises very well, is the presence of distinct, large hooks running in two series along the oral surface of the animal (Engeser & Clarke, 1988). Figs.10 and 11 show photographs of fossilised belemnoid remains.
The basic belemnoid hook probably comprised the same components as that of extant coleoids; namely chitin. It is possible that the hooks contained more components, such as harder, possibly mineral, materials, but then the only evidence for this really is that the hooks are so well preserved, compared to fossil neocoleoid suckers rings. The hook was positioned curving inwards, and in two series running along each arm, in pairs of between 20 and 50.

fig. 10: Acanthoteuthis from Solenhofen, Germany, currently residing in the collection of U. S. National Museum of Natural History. Of particular recognition is the clear detail of the animal's body, including the hooks clearly radiating off the arms!
fig. 11: belemnoid hooks (possibly Belemnoteuthis) taken from Lyme Regis in Dorset, England, also in the collection of U. S. National Museum of Natural History. Both photographs used with kind permission by Richard E. Young, University of Hawaii.
The hook itself is split up into several sections, each with its own distinct nomenclature as shown in fig.13. The presence of this small hairline groove known as the orbicular scar indicates that the hook was probably not exposed, and was housed within a sheath of soft tissue. Because the tissue attachment point is quite far up the shaft, only the uncinus would be visible, or at least naked. Engeser and Clarke proposed that the hook was stalked, and that it was attached by muscles that could have allowed lateral movement. Since the belemnoid did not have an extensile pair of limbs, like the tentacles of squids and cuttlefish, or the flexible, muscular sucker discs like octopuses, it would have had to manipulate the prey for eating by using its whole arm. If the hooks were sessile, bearing in mind that it may have had up to fifty, then the belemnoid would be severely limited in its arms' movement. Having hooks that could be moved, even slightly, would allow the animal more movement. The manner in which the hooks were positioned, recurved, would have also helped, in that the trapped prey, which was obviously caught by being impaled, could only move in one direction in order to free itself from the hook: towards the mouth. According to Engeser and Clarke, there are three groups of belemnoids that display arm hooks, and one that does not. The Phragmoteuthids are reported as far back as the late Permian and are described as having more slender, slighter hooks than what is described as the "normal" or archetypal hook (fig.13) although variations including enlarged bases and more curved uncini are reported in species. The Belemnoteuthids have more typical hooks, although Chondroteuthis wunnenbergi Bode 1933, exhibited prominent spurs on the proximal lower third. The Belemnitids, most well known as the belemnites, also display a deal of variation. Acrocoelites (Toaricibelus) raui Werner 1912 appears to have two different types of hook, differentiating from the typical hook to those with an internal spur towards the distal end of the arm. Two fossil specimens of Passaloteuthis paxillosa Schlotheim 1820 showed the same structural oddities of having no hooks whatsoever towards the distal parts of the arms. This suggests that this species may well have lacked hooks at the ends of their arms. Unfortunately no other specimens were described or even mentioned, so a conclusion can only be drawn from these two fossils alone.
fig. 13: schematic of a typical belemnoid hook, adapted from Engeser & Clarke
Ideally a more thorough investigation into prehistoric cephalopod arm structure would have been desirable, but there is so little fossil evidence of cephalopods, being a soft-bodied animal naturally, that such a task is nigh on impossible at present. Most of the neocoleoid fossils are little more than faint outlines with maybe the presence of one or two organs, or surfaces, but this can be sketchy at best, since the fossils are interpreted differently. For this reason, only belemnoids have been included as a representative of prehistoric cephalopods since the available information on fossil cephalopods with regard to armature is pretty much restricted to the belemnoids. It is curious just how well these animals have preserved compared to the other prehistoric cephalopods, given that this class has been in the world's oceans since the Late Cambrian.
SPECIAL CASES
The case of Stauroteuthis syrtensis:
Stauroteuthis syrtensis Verrill 1879 (Johnsen et al., 1999) is a deep sea cirrate octopod that sports very unique armature. It is similar morphologically to other cirrate octopods in that it possesses two rows of cirri along the oral surface (the underside) of each arm, and also has a long web that connects each arm with a single series of structures analogous to the suckers of a typical octopus such as E. cirrhosa or Octopus vulgaris. However, these structures only vaguely resemble their adhesive counterparts, and can even be described as vestigial. Indeed these suckers no longer perform any adhesive role whatsoever, instead they exhibit Bioluminescence! Although bioluminescence is common among the decapods (Johnsen et al.) it is incredibly rare in octopods, being described in that manuscript as only being present in "2 of the 43 genera", and even then only found in breeding females. In this species, however, it appears that both females and males possess the light organs (photophores). Robison and Young (1981) observed that the light emitted from these modified suckers were of a definite green hue, compared to the blue-green light more commonly found in bioluminescent animals. This does set it apart from other inhabitants of the deep with such similar abilities!
The problem with the information regarding this species, as pointed out by the team that produced said document, is that it lives at great depths. Indeed the three specimens were caught in a range of 750- 920m using deep trawling methods. They are understandably difficult to study in their natural environment not only due to the depth, or the mind-blowing vastness of its habitat, but also because the octopod can probably detect the submersible light long before contact, meaning that it is unclear whether the behaviour observed is natural or induced by the presence of the submersible. It is described as hanging in the water column with its arms and web outstretched to create a sort of bell-shaped posture. It is possible that this is a threat response, but it appears to be a natural position rather than that of defence. There are a few theories as to why such an animal would develop bioluminescence, which can be both an advantage and a disadvantage, as will be discussed.
S. syrtensis exhibits sexual dimorphism in that mature males have grossly enlarged suckers (or photophores) compared to those of a female of similar development. Since both have the bioluminescent ability, it was proposed that it was used as sexual displays. Sexual signalling is understandably common in a phylum that appears to rely so much on vision, and so this theory would appear to make sense. The evidence of a distinct light colour also enables what Johnsen, Balser, Fisher and Widder described as a "private line of communication" between members of the same species. Indeed, any clear visual cue that distinguishes you from another species, especially in the dark, is bound to be favourable! However, there are a few factors that may serve to contradict this theory. The first is that in this instance, all three specimens examined had these photophores, and yet they all appeared to be immature. Surely only mature individuals, with the ability to reproduce, would only need these? But then, we only know a small amount about this species, so anything is possible. Another problem is that the octoradial pattern (imagine eight twinkling spokes) would be distorted after a certain distance due to light dispersal. However, it is questionable that this would prove too much a problem since the photophores emit a fairly distinct green colour! Finally this colour would be clearly visible to potential predators. That said, although in a slightly different context, many male birds compromise cryptic camouflage in favour of conspicuous coloration and displays that give its presence away, in order to attract mates. Although sexual signalling probably isn't the primary reason for photophores, I am not, at such an early stage, wholly willing to dispel the possibility.
This being the case, a slightly more plausible hypothesis describes the light's use in prey capture. S. syrtensis feeds on small, planktonic crustaceans. It has been proposed that cirrate octopods secrete lines of mucous web which are handled by the cirri to trap the animal's prey. This kind of method has been suggested for three of the cirrate genera (all of which possess "non-functional" suckers), Stauroteuthis being one of them. In this case, the bell posture could be easily explained. It is possible, therefore, that the photophores are used as a lure, similar to the angler fish. These crustaceans have well developed eyes, and seem to be attracted to light sources, which would serve the S. syrtensis very well. The fact that the animal is usually observed in the bell or umbrella posture either oriented with its mouth facing upwards, or facing downwards could coincide with nutrient flows in the water column, since many planktonic and nektonic animals make vertical migrations every night in order to feed on phytoplankton. On the other hand it could just be, as Denton (1990) stated, because upwelling light intensity is a small percentage of downwelling light intensity, which Johnsen et al. proclaimed would make the octopod highly visible to potential prey higher up in the water column, although this statement I have difficulty in imagining. Surely if the light intensity of an octopod facing upwards was less it would be less visible to prey above it?
The case of the Vampyromoprh:
The Vampire squid, Vampyroteuthis infernalis Chun 1903 is the only known remaining member of the order Vampyromorpha (sometimes Vampyromorphida). There are fossils of these animals ranging back to the Mesozoic, which is rather remarkable given their gelatinous tissue composition! A resident of the deep oceans, this is yet another relatively unknown animal.
The vampire squid, although commonly referred to as a "squid", is a member of the Octopodiforme superorder. Its second arms (Arms 2; the ones octopods have lost) have been modified to retractile sensory filaments that actually look like they are not part of the limb structure since they are not connected by the web. Like the cirrates they only possess one series of suckers, which are "banked" by a row of cirri on either side, and in V. infernalis are only present of the distal half. The web is attached to the arms to about two thirds of the way.
Young (1967) showed that the filaments are innervated differently to the other eight arms, which suggests that they are derived from a source different to the "decapod ancestor". These filaments are used in feeding and are probably used like a fishing line. A prey item bumps into the filament in the dark waters. Upon sensing this stimulus, the vampire squid lunges toward the source in the hope of enveloping it with the arms and web. It is possible that the cirri play a part in this too. The vampire squid also displays unique behaviours, especially in response to a potential threat. One threat response is to invert its arms and web round its head, as if it's turning itself "inside out", and extending its cirri outwards so that they resemble spikes or hooks (fig.14) in what is known as a "Pineapple posture".
fig. 14: photograph of the vampire squid in its "pineapple" posture with cirri held rigid.
Another predator-avoidance strategy is the use of photophores in the skin, combined with ejection of mucous containing bubbles of bioluminescent light. Once a string has been completed the squid halts operation of its photophores and slips of, leaving the bioluminescent mucous thread decoy with the predator.
The suckers of V. infernalis lack a cuticular lining, which may suggest that they do not have the ability to seal the sucker to an object, meaning the sucker would be useless, if not extremely weak, as an adhesive structure.
The case of the Nautiluses:
The nautiloids are the oldest cephalopods on the planet. From the Cambrian to the Early Devonian they were the only cephalopods found in the oceans, and even continued to dominate right through to the Jurassic period, where they experienced a sharp decline, which saw the extinction of the orthoconic nautiloids. It is the early nautilda that gave rise to our modern nautiluses, of which there are only around six known species. The early nautilda showed many of the characteristics familiar with these modern species. The nautilus really is a relic of an ancient time!
What sets the nautiluses apart from the coleoid cephalopods is the absence of suckers. By definition, nautiluses do not have arms, and instead can have anywhere up to ninety tentacles which are kept in sheaths when not in use. The absence of suckers does not dictate a lack of adhesive ability however, and the nautilus still employs special structures to allow it to grip objects, albeit using a completely different strategy.
Nautiluses have, lining their tentacles, a series of alternating ridges, or cirri, and grooves. These allow nautiluses to grip substrata or prey with an effect similar to Velcro. In the emperor nautilus, Nautilus pompilius Linnaeus 1758, and most likely in the other five species (and both N. pompilius subspecies), the cirri are in turn lined with annular ridges that run round the circumference of each cirrus, and are more pronounced on the oral side (Muntz & Wentworth, 1995). The epithelium exhibits thickening on the proximal surface of these ridges, and houses electron-dense granules which are only found in this location on the cirrus. The granules contain a mucopolysaccharide which could be what facilitates adhesion.
Nautiluses have incredibly poor vision; they're eyes being little more than pinhole eyes (Basil et al., 2000), meaning that they can probably only distinguish light and dark, and are most likely unable to clearly define objects. Clearly nautiluses do not rely on vision to hunt or scavenge; they tend to feed on dead matter but they are still active predators so they need to forage, and locate prey.
Their tentacles also house cells that resemble taste buds (Basil et al.), but were found not to allow the animal to track the odour, or chemical signal. For this the animal requires two specialised organs located below the eye called Rhinophores. While the tentacles were found to pick up chemicals in the water, it was the rhinophores that allowed them to locate the source. Test animals with their rhinophores temporarily plugged did show a distinct behavioural change upon the presence of a chemical in the water, but they could not effectively locate the source. This suggests that the tentacles could pick up preliminary stimuli which tell the animal that a food source is there. The emperor nautilus inhabits fairly deep regions (300m approx.) and migrates vertically at night to feed. Essentially it lives in a dimly-lit, or even completely dark, world. If the animal was using its rhinophores all the time, such a behaviour would surely be wasteful, and so a preliminary warning system that tells the animal when it can use its rhinophores to locate prey would be a huge benefit.
Aside from foraging, the nautilus also uses its tentacles to adhere to surfaces during stages of inactivity. According to O'Dor, 2002, nautiluses engage in a state of torpor akin to sleep. He also stated that an animal in this state, if not anchored to any substratum, would drift around at a velocity fairly close to its optimum speed, in a random fashion. This movement is induced by breathing alone. Since nautiluses achieve locomotion by "jetting". Since they move by expelling water from their mantle cavity, the same site where the gills are situated, random movement would be understandable during unconsciousness. This of course poses many hazardous implications for the nautilus, and the animal would quite rightly want to stay put!
CONCLUSION
Similarities and Differences:
The sucker of the decapods and octopods are analogous. They both perform exactly the same function; adherence to a surface, but the morphology is completely different. The basic uses are also the same, although the octopods tend to have more uses for their sucker pads.
It would appear that decapod suckers are very limited in their uses, being mostly for prey capture: to aid in the adherence of the sucker to the body of the prey, and in some cases, probably by impalement! Some hook structures, such as those found in sexually dimorphic species, especially in those in which the dimorphism is dictated by the hooks, there is the possibility of a use in reproduction, most likely in species that reproduce through direct implantation by the male of sperm into the female's mantle cavity. Aside from this, decapod suckers do not appear to have any other role. Octopuses use their suckers for more than just prey capture and manipulation, and the suckers are more flexible and muscular. The sucker structure itself is relatively sessile, but the rim of the infundibulum has a remarkable array of movement. Octopuses are regarded as highly intelligent animals, which could be a result of their multipurpose suckers, or even vice versa. The intelligence of decapods was not researched for this paper, but it is possible that squids at least cannot achieve the intellectual level of an octopus, on the evidence of their highly specific armature. This statement is completely subjective, however, but it is one conclusion that can be drawn from the observation of the different structures.
The decapods do show a lot more variety in terms of structure, such as the shape and size of the ring in relation to the sucker. This may be a reflection on the animal's prey. Since nearly every family, at least, have architectural differences in the ring the different shapes could prove optimal formations for catching particular prey species, or perform better at different velocities or depths. In a three-dimensional world, it would also be wise to include the decapod's angle in relation it its prey, which might also affect the success of the tentacle strike. Decapods typically have a very fast tentacle strike; the tentacle extending and contacting a prey item in about 15-35ms (Smith, referring to Kier, 1982), with an incredibly high success rate: normally 80-90%. The chitinous rings could play a large part in this success, allowing an initial grip which facilitates formation of a rim, in turn allowing adhesion by pressure differential. Owing to the highly specialised use of tentacles, it is possible that decapods do not have as much motor control over their tentacles as they do their arms.
Octopus capture methods may differ somewhat as the octopus is using one or more of its arms to grab a prey item, which it probably has more control over, compared to a pair of tentacles. As such it can probably allow for a much surer strike. Octopus arms also frequently coil up when they grab an object, which no doubt increases its hold over the item, which means that they may not require hard, claw-like formations. Octopuses also lunge at their prey with all their arms, usually from above, in the water column. This behaviour is also demonstrated by cuttlefish. Another reason why octopuses do not require hooks or rings is that they are ambush predators and are masters of camouflage, able to change their appearance and colour at will. This means that they can close in on a prey animal, and will only strike when they are sure of success. In a three-dimentional environment, such as those of pelagic squids, there is no such luxury of a complete surprise attack, and so the squid would have to rely more on a high-velocity mode of capture.
Indeed, hard parts may even prove detrimental to an octopus, given the majority of octopuses' habitats; the sea floor. Octopuses live in and around rocks, and frequently reside in dens. It is common knowledge of the octopus' ability to squeeze into the narrowest gaps, and its inquisitiveness and tendency to explore and probe. An octopus can go anywhere so long as its beak can get through. If an octopus was carrying around so many hard structures in its arms, then it would be severely limited in where it would go.
It seems to be the case generalisation demands a simpler anatomical structure (in this case the octopod's suckers) whereas specialisation begins to limit the range of uses, in order for that structure to be optimal in that one special activity. This appears to also be the case in cephalopods with regards to their structures.
Cephalopods are very visually oriented animals, and employ the use of complex signals. This does include very complex, and impressive colour displays using chromatophores to switch to a variety of colours to beautiful effect, but the use of arm signals is also important. Although this is more holistic, and may not even include the suckers in some cases, an interesting implication does arise when considering sexual dimorphism. As in the case of L. grimaldii it was presumed that the hooks were used physically in mating, but just as probable is role in display. Just as stags sport their impressive antlers during the rut, that are usually only employed as a grappling weapon if two competing males refuse to back down, so the male scaled squid may perform similar displays to impress a female. Until two scaled squid are found in the act of mating, we can only speculate.
A conclusion can also be drawn from the terminology derived from the observation of cephalopod limbs, namely the tentacles. It is found that in both the coleoid decapods and the nautilus both have limbs called tentacles. Which it was observed that tentacles were specialised limbs, what is consistent in both subclasses is that these limbs are fully retractable and extendible. Indeed nautiluses only sport this kind of limb, and do not have any unspecialised arm. Whether it was by coincidence, it can nevertheless possible that the term tentacle can refer to a limb which is extendable.
A possible Evolutionary Theory:
The existing Nautiloidea, of the genus Nautilus, exhibit what could be classed as a rather primitive array of armature. Providing the Nautiloidea did possess these adhesive ridges to begin with, and it wasn't just convergent evolution displayed by the nautiluses, this could very well explain the beginnings of the cephalopod sucker structure. It's doubtful that nautiluses alone would just happen to develop an adhesive system on their limbs in recent times without good reason, and so it would be expected that the ridges are the very ancestral progenitor of the sucker pad. This theory would be ideally backed up by the discovery of soft-bodied bactritid ammonoid fossils, these being the intermediary stage between nautiloidea and coleoidea, and ancestral to the later ammonoids with perfectly preserved arms that could show whether any structures were present. Since in ammonoids the only fossils reported have thus far only been the shell, conclusions have to be drawn.
Assuming that the nautiloids did have adhesive ridges, it can be suggested that these were carried and further developed by the ammonoidea: more specifically the aforementioned bactritids, which gave rise to the coleoids.
Assuming also that the bactritids (at least) had some sort of ancestral structure, such as a hook, makes the presence of the belemnoid hook more plausible as well as the neocoleoid hook. These structures could not have just sprung up out of nowhere.
It is possible, therefore, that the hook structure was possessed by a common ancestor to belemnoids and neocoleoids. This common ancestor could have had an ancestral hook. When the belemnoids and neocoleoids diverged, the belemnoids may have kept the ancestral hook (or indeed developed it in a completely different way!), while the neocoleoids developed the hook into a sucker pad. A short diagram showing the development from hook to sucker is shown in fig. 15.
fig. 15: a depicts development from an ancestral hook similar to that of a belemnoid's to that of a modern decapod, including the formation of the cavity (acetabulum) and a stalk, and the transition from a single hook to a ring of denticles. Included is b which depicts what could happen, carrying on from a in the same direction.
For sake of completeness, further development into the octopods sucker, lacking a ring, is included. If this were to happen, then because octopods did not branch off of decapods, and both more or less diverged in the same manner as with the belemnoids, theoretically the first octopods would have had some hard structures until these vanished. Of course the internal morphology would have to change again, but slight modification of the infundibulum and acetabulum, and the development of the piston into the acetabular roof could facilitate this. This is highly speculative as there is not a lot of evidence of fossil neocoleoids, but I'd like to hope that it fits together as a sound theory. One interesting fact is that the "paper nautilus" Argonaut argo, an incirrate octopus that secretes a very thin shell, appears to have hooks between the rim of the sicker and infundibulum (Nixon & Dilly); could this be a throwback to a day when all coleoid cephalopods had hard armature?
Thanks:
I would like to thank the following people for their advice and help throughout the duration of my study, in no particular order:
I thank the display manager at St. Andrews Aquarium for the specimen of Eledone cirrhosa as well as giving advice and showing an interest in the study. I also thank William Kier for providing me with his two papers on octopus suckers and having an interest from the offset.
Special thanks goes to the staff and forum members of TONMO, for the endless help, ideas, advice and critical views, including Phil Eyden for proof reading this paper and providing extensive information on fossil cephalopods, Dr Steve O'Shea of Auckland University of Technology in New Zealand, and especially Kat Bolstad, also of Auckland University, who provided endless help and sources. I would have struggled to obtain some sources otherwise.
Finally, within the department, I would also like to thank several people. Obviously I would like to thank my supervisor, Dr Allan Jones, for suggesting the topic that has dominated my life for the last half year, as well as providing valuable advice and moral support. Also of note is the university library, who managed to negotiate an extended inter-library loan of "Cephalopods of the World" for me. I would also like to thank a fellow student, Peter Rendle, for posing very interesting questions, as well as good, in-depth discussion regarding the subject.
REFERENCES
Anderson F. E. 2000, Phylogenetic relationships among loliginid squids (Cephalopoda: Myopsidia) based on analyses of multiple data sets. Zoological Journal of the Linnean Society 130: 603-633
Bolstad K. S., O'Shea S. 2004, Gut contents of a giant squid Architeuthis dux (cephalopoda: Oegopsida) from New Zealand waters. New Zealand Journal of Zoology 31: 15-21
Chase r., Wells M. J. 1986, Chemotactic behaviour in Octopus. Journal of Comparative Physiology 158/3: 375-381
Christensen W. K. 2002, Fusiteuthis polonica, a rare and unusual belemnite from the Maastrichtian. Acta Palaeontologica Polonica 47: 679-283
Dzik J. 1980, Origin of the cephalopoda. Acta Palaeontologica Polonica 26: 161-191
Engeser T. S., Clarke M. R. 1988, Cephalopod hook, both recent and fossil. The Mollusca 12: 133-151
Fiorito G., Gherardi F. 1999, Prey-handling of Octopus vulgaris (Mollusca, Cephalopoda) on Bivalve preys. Bejhavioural Processes 46: 75-88
Förch E. C., Uozumi Y. 1990, Discovery of a specimen of Lycoteithis lorigera (Steenstrup, 1875) (Cephalopoda: Teuthiodea) from New Zealand and Additional notes on its morphology. New Zealand Journal of Marine and Freshwater Research 24: 251-258
Jackson G. D., O'Shea S. 2003, Unique hooks in the male scaled squid Lepidoteuthis grimaldii Joubin 1895. Unpublished but available through the TONMO website
Johnsen S., Balser E. J., Fisher E. C., Widder E. A. 1999, Bioluminescence in the deep-sea cirrate octopod Stauroteuthis syrtensis (Mollusca: Cephalopoda). Biological Bulletin 197: 26-39
Keir W. M., Smith A. M. 1990, The morphology and mechanics of octopus suckers. Biological Bulletin 178: 126-136
Keir W. M., Smith A. M. 2002, The structure and adhesive mechanism of octopus suckers. Integrated Comparative Biology 42: 1146-1153
Kier W. M., Van Leeuwen J. L. 1997, A kinematic analysis of tentacle extension in the squid Loligo pealei. The Journal of Experimental Biology 200: 41-53
Lindgren A. R., Giribet G., Nishiguchi M. K. 2004, A combined approach to the phylogeny of Cephalopoda (Mollusca). Cladistics 20: 454-486
Lipinski M. R. 2001, Preliminary description of two new species of cephalopods (Cephalopoda: Brachioteuthidae) from South Atlantic and Antarctic waters. Bulletin of the Sea Fisheries Institute 1 (152) : 3-14
Lukeneder A. 2005, First nearly complete skeleton of the Cretaceous duvaliid belemnite Conobelus. Acta Geologica Polonica 2: 147-162
Muntz W. R. A., Wentworth S. L. 1995, Structure of the adhesive surface of the digital tentacles of Nautilus pompilius. Journal of the Marine Boilogical Association of the United Kindom 75 (3): 747-750
Nixon M., Dilly P. N. 1977, Sucker surfaces and prey capture. Symposium, Zoological Society, London 38: 447-511
Robison B. H., Young R. E. 1981, Bioluminescence in pelagic octopods. Pac. Sci. 35: 39-44
Smith A. M. 1996, Cephalopod sucker design and the physical limits to negative pressure. The Journal of Experimental Biology 199: 949-958
Vecchione M., Galbraith J. 2001, Cephalopod species collected by deepwater exploratory fishing off New England. Fisheries Research 51: 385-391
Nesis K. 1987, Cephalopods of the World. Originally published in Russia, 1982, this translated edition T.F.H Publications ISBN 0-86622-051-8
Tree of Life web project: www.tolweb.org/tree
Cephbase: www.cephbase.utmb.edu
Waikiki Aquarium website: www.waquarium.org
The Cephalopod Page: www.thecephalopodpage.org
Paleos: www.palaeos.com
All sites are operational as of July 2007.